
The debate over whether the UK’s National Health Service should embrace magic mushrooms and other psychedelic compounds for treating severe mental health conditions like depression is intensifying, pitting the potential for groundbreaking new therapies against deep-seated caution regarding these historically illicit substances. Science Correspondent Pallab Ghosh delves into this complex issue, exploring the hopes of those who have found relief, the warnings from those who have suffered adverse effects, and the scientific and regulatory hurdles standing in the way of wider adoption.

The landscape of mental health treatment in the UK, and indeed globally, is fraught with challenges. Many patients struggle to find effective long-term solutions, and conventional antidepressants, while vital for many, don’t work for everyone and often come with significant side effects and slow onset of action. This urgent need for innovative approaches has rekindled interest in psychedelics, once relegated to the fringes of medicine.
Larissa Hope, a former actress who appeared in the TV drama Skins, stands as a compelling advocate for the therapeutic potential of psilocybin, the active ingredient in magic mushrooms. Her early career success inadvertently unearthed a buried trauma, plunging her into a severe mental health crisis. Conventional antidepressants, she recalls, offered little solace, failing to address the root causes of her distress or alleviate her persistent suicidal ideation. A pivotal moment arrived when she undertook a small dose of psilocybin, administered under strict clinical supervision. She describes the experience as profoundly transformative. "When I experienced it, I burst out crying," she recounts today, her voice still imbued with the memory of that release. "It was the first time in my life I had ever felt a sense of belonging and safety in my body. I kept saying, ‘I’m home, I’m home’." Nearly two decades later, Larissa firmly believes that this single, carefully managed psilocybin session, coupled with ongoing therapy, was instrumental in helping her confront and ultimately overcome her suicidal feelings, offering a profound sense of peace and integration she had long sought.

However, not everyone’s journey with psychedelics mirrors Larissa’s positive outcome. Jules Evans, a respected university researcher, experienced a starkly different and deeply troubling encounter with LSD during his youth, albeit for recreational purposes, at the age of 18. His trip spiraled into what he vividly describes as a "deluded" state, marked by intense paranoia and a terrifying sense of self-disintegration. "I believed that everyone was talking about me, criticising me, judging me," he remembers, the fear still palpable in his words. "I thought, I’ve permanently damaged myself; I’ve permanently lost my mind. It was the most terrifying experience of my life." The repercussions were long-lasting; he grappled with severe social anxiety and panic attacks for years, ultimately receiving a diagnosis of post-traumatic stress disorder (PTSD). Today, Evans channels his experience into positive action as the director of the Challenging Psychedelic Experiences Project, an initiative dedicated to supporting individuals who encounter difficulties after taking psychedelics. His work highlights the critical need for understanding and mitigating the potential risks associated with these powerful substances.
These two contrasting narratives—one of profound healing, the other of enduring trauma—underscore the complex dilemma now confronting medical professionals, regulatory bodies, and policymakers. The central question remains: should doctors be empowered to prescribe treatments that incorporate magic mushrooms and other potentially beneficial psychedelic drugs, integrating them into mainstream healthcare systems like the NHS?

The resurgence of interest in psychedelic medicine is driven by a growing body of new scientific studies suggesting that these compounds could offer significant therapeutic benefits for a range of mental health conditions. Beyond depression, researchers are exploring their efficacy in treating obsessive-compulsive disorder (OCD), various forms of PTSD, complex trauma, and even severe addictions to substances like alcohol and gambling. Currently, the use of psychedelic medicine outside of authorized research or tightly controlled clinical trials remains illegal in the UK. Yet, since 2022, an increasing number of such trials—more than 20—have been initiated to investigate different psychedelic medicines for these debilitating conditions. While many of these studies have yielded promising results, suggesting that these treatments can indeed help, others have produced mixed or unclear findings. Crucially, only a small minority have definitively found no benefit on their primary outcome measures.
A highly anticipated moment in this unfolding narrative is the expected release of results later this year from one of the largest clinical trials into psilocybin’s use, conducted by the UK biotech firm Compass Pathways. The UK’s medicines regulator is keenly awaiting this data, as it will be a critical factor in their consideration of whether to ease current stringent restrictions and potentially allow the use of psychedelic medicine beyond research and trials.
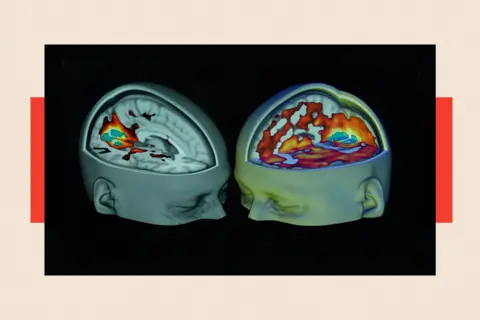
Professor Oliver Howes, who chairs the Royal College of Psychiatrists’ Psychopharmacology Committee, expresses cautious optimism. He views psychedelics as a "promising potential new treatment for psychiatric disorders," including for patients within the NHS. "One of the key messages is that this is something we desperately need – more treatments and better treatments for mental health disorders," he states, emphasizing the urgency. He highlights the "really interesting" promise shown in smaller-scale studies and the potential for these treatments to "work quicker" than conventional methods. However, Professor Howes also stresses the paramount importance of evidence and warns against "overhyping the potential benefits" before comprehensive trial data is available. His caution is echoed by a report from the Royal College of Psychiatrists, published in September 2025 (presumably a prospective or very recent report), which warned of the potential dangers associated with psychedelics. Doctors consistently emphasize that recreational use of psychedelic drugs is not only illegal but can also be profoundly harmful.
The therapeutic use of psychoactive substances is, in fact, as old as civilization itself, with magic mushrooms, opium, and cannabis having been integral to both recreational activities and spiritual rituals for millennia. However, the modern era saw a dramatic shift. By the 1960s and 1970s, LSD, colloquially known as acid, became deeply intertwined with the burgeoning counterculture movement, famously championed by Harvard psychologist and counterculture guru, Timothy Leary. His iconic exhortation to young people to "turn on, tune in, drop out" encapsulated a philosophy of awakening inner potential, engaging with societal critique, and disengaging from conventional norms. This period, however, also saw these drugs increasingly associated with social unrest and perceived moral decline. Consequently, by the late 1960s and early 1970s, strict bans were implemented globally, leading to greater restrictions on scientific research into their potential medical applications, effectively halting progress for decades.

This long scientific dormancy began to lift in the 2010s, thanks to a series of ground-breaking scientific developments led by Professor David Nutt and his pioneering team at Imperial College London. Their subsequent clinical trials on patients suffering from depression provided compelling evidence that psilocybin was at least as effective as conventional antidepressants, and critically, appeared to do so with fewer debilitating side effects. Professor Nutt also highlighted another significant advantage: the remarkable speed of its action. "We thought rather than wait for eight weeks for antidepressants to switch off the part of the brain associated with depression, maybe psilocybin could switch it off in the space of a few minutes," he posited, suggesting a rapid neural reset.
While scientifically promising, Professor Nutt’s assertions have not been without controversy. His outspoken views led to his dismissal in 2009 from his position as chair of the government’s drugs advisory body, the Advisory Committee on the Misuse of Drugs (ACMD), by the then Labour Home Secretary Alan Johnson. Public comments, such as his assertion that there was "not much difference" between the harm caused by horse-riding and ecstasy, were deemed incompatible with his role as a government adviser, sparking a wider debate about scientific freedom and public policy. Nevertheless, Professor Nutt’s pioneering studies undeniably reignited global interest, sparking numerous investigations into the therapeutic benefits of various other psychedelic drugs.

The crucial question remains: should these powerful tools truly be made available on the NHS? At University College London, neuroscientist Dr. Ravi Das is at the forefront of this inquiry, attempting to unravel the complex mechanisms by which certain habits become ingrained addictions while others fade. He believes psychedelics may hold a key to unlocking these answers. Dr. Das is currently leading a study recruiting heavy drinkers to investigate whether dimethyltryptamine (DMT), a short-acting psychedelic also known for recreational use, can effectively target the brain’s memory and learning systems. This research builds upon existing evidence suggesting that psilocybin has the capacity to disrupt the deeply entrenched habitual behaviors associated with addiction.
"Every time someone drinks, a bit like Pavlov’s dog, you’re learning to associate things in the environment with the rewarding effect of alcohol," Dr. Das explains. "We’ve been focusing on whether certain drugs, such as psychedelics, can break down those associations." While this study is still in its very early stages, its ultimate aim, if successful in both this and future trials, is to secure regulatory approval and offer it as a treatment option within the NHS. Dr. Das firmly believes in equitable access: "If psychedelic therapies prove to be both safe and more effective than current treatments, I would hope to see them made accessible via the NHS – rather than to just the privileged few who can afford them privately."

The legal status of these substances is a significant barrier. Ketamine, which was the subject of a previous trial by Dr. Das and is increasingly used in mental health treatment, occupies a different legal category and can be used medically in the UK. However, other psychedelics such as DMT, LSD, psilocybin, and MDMA are currently classified as Schedule 1 drugs, meaning they are deemed to have no legitimate medical use. This classification places them under the tightest controls, severely limiting their use even for research and requiring extremely difficult-to-obtain medical licenses. Dr. Das remains hopeful that mounting positive scientific evidence from trials will ultimately compel the government to revise the scheduling of these drugs.
However, a critical analysis published in the British Medical Journal in November 2024 by PhD student Cédric Lemarchand and his colleagues raised important questions about the ease of precisely determining the effects of psychedelic drugs. They noted, "Because hallucinogens are often combined with a psychotherapy component, it is difficult to separate the effects of the drug from the therapeutic context, complicating comprehensive evaluations and product labelling." The analysis also warned that short-term trials might not adequately detect "the potential for harm and serious adverse events from long-term use of hallucinogens… The potential for abuse or misuse must also be considered." These points highlight the complexities of rigorous scientific validation in this field.

While research continues to suggest the therapeutic benefits of psychedelic medicines, medical professionals like Professor Howes maintain a cautious stance. He argues that, with the exception of ketamine (which has undergone regulatory assessment), psychedelic treatments should not become routine medical practice in the UK outside of controlled research settings until larger, more rigorous trials provide robust evidence of their safety and effectiveness. "In a clinical trial setting, it’s very carefully evaluated," he emphasizes. "If people take these on their own or in a backstreet clinic, then there is no guarantee of that, and the safety issues start becoming a major issue."
His warnings are substantiated by alarming figures gathered by the Challenging Psychedelic Experiences Project. Their data suggests that a significant 52% of respondents who regularly use psychedelic drugs reported having an intensely challenging psychedelic trip, with 39% of those considering it "one of the five most difficult experiences of their life." Furthermore, 6.7% reported considering harming themselves or others following a challenging experience, and 8.9% reported being "impaired" for more than a day after a difficult trip. Jules Evans notes that some individuals even required medical or psychiatric assistance and continued to feel worse for weeks, months, or even years after their adverse experiences. "Ideally, I would love doctors and regulators to know more about these adverse effects, and how people can recover from them, before they say, any of these therapies are safe," he argues passionately.

Despite these legitimate concerns, proponents like Professor Nutt, Professor Howes, and Dr. Das argue that progress in bringing these promising treatments into clinical practice is being unnecessarily hampered by the bureaucratic labyrinth involved in obtaining permission for medically supervised clinical trials. "There are so many people suffering unnecessarily," Professor Nutt lamented to BBC News. "And some of them are dying, because of the unreasonable barriers to research and treatment that we face in this country. It is, in my view, a moral failing." He strongly advocates that "When these medicines are proven to be safe and effective, I think it is vital they are made available through the NHS to all who need them, not limited to the private sector, as happened with medical cannabis." Professor Howes, while urging caution, shares this sentiment regarding research barriers: "There are big barriers to doing this research, so we do ask for the government to review the regulations of these substances, for research, because it does lead to long delays, and, we desperately do need new treatments."
The analysis from Mr. Lemarchand and his colleagues underscores the need for greater scrutiny of trials. They call for medical journals to "appraise the evidence more critically, fully account for limitations, avoid spin and unsubstantiated claims, and correct the record when needed" to ensure that hallucinogens are rigorously vetted before being endorsed as safe and effective treatments. The Advisory Council on the Misuse of Drugs maintains its blunt stance that Schedule 1 drugs "contain those of no medicinal value," justifying the tightest controls, with ministers directly linking the Home Office licensing regime to public protection.

In response, the government has backed plans to ease licensing requirements for certain clinical trials approved by the Medicines and Healthcare products Regulatory Agency (MHRA) and the Health Research Authority (HRA). Work is currently underway to implement exemptions for specific universities and NHS sites, with a cross-government working group coordinating a cautious rollout pending the results of pilot projects. However, some doctors, including Professor Howes, contend that these changes are progressing "painfully slowly," with "a lot of red tape holding things up."
Supporters of psychedelic medicines hold out hope that the upcoming phase three trials by Compass Pathways will pave the way for further relaxations, at least concerning research. For Larissa Hope, the potential is undeniable. She firmly believes her experience with psilocybin, the active ingredient in magic mushrooms, provided profound insight into her suicidal ideation and trauma. "I had a solid plan to end my life. Then suddenly, death wasn’t the only way," she reflects. "Under psilocybin, my nervous system began, for the first time, to recognise what peace felt like." Her testimony, alongside the accelerating scientific inquiry, ensures that the debate over psychedelics in the NHS will continue to be a vital one.








