
Amanda James-Hammett was a vibrant 37-year-old when a sudden, catastrophic event struck while she was engaged in the mundane task of doing the dishes at home. The sensation was jarring, described by Amanda as "a pop in my head, like a big bubble." In the terrifying moments that followed, her attempt to cry out morphed into an incoherent sound, a chilling testament to the fact that "something wasn’t right." Within hours, the life she knew was irrevocably altered as she lost the ability to speak and control her right arm. The arduous journey of relearning fundamental skills – speaking, reading, and performing everyday actions – had begun. Six years on, Amanda is a participant in a groundbreaking nationwide NHS trial, testing a novel "at home" device designed to empower stroke survivors in regaining crucial hand and arm movement. Her sentiment is profound: "It’s about freedom. It’s given me my freedom back."
This pioneering technology is currently being rigorously evaluated at 19 NHS sites across the United Kingdom, spearheaded by the Sheffield Teaching Hospitals NHS Foundation Trust in collaboration with the University of Sheffield. The researchers behind this ambitious initiative, dubbed the Triceps trial, have secured £2 million in funding, marking it as the most extensive trial of brain stimulation and stroke rehabilitation ever undertaken. The trial aims to enrol approximately 270 individuals, offering a beacon of hope for those affected by this debilitating condition.
Dr. Sheharyar Baig, a distinguished neurologist at Sheffield’s Royal Hallamshire Hospital, provides crucial context: "Stroke is an interruption to the blood supply to the brain. When the blood supply is interrupted, unfortunately, the brain stops functioning in a certain area. That can lead to all manner of symptoms, from weakness to visual problems and speech problems." He strongly advises anyone experiencing such symptoms to call 999 immediately.

The statistics surrounding stroke are stark and underscore the urgent need for innovative rehabilitation strategies. Dr. Baig highlights that stroke is "the leading cause of adult-onset disability in the UK," impacting an estimated 100,000 people annually. The long-term consequences ripple through society, with over a million people currently living with its enduring effects. A significant proportion, around half of all stroke survivors, grapple with ongoing arm weakness, a condition that can manifest as a subtle loss of dexterity or a complete absence of movement. This impairment profoundly affects daily life, making even simple tasks like dressing, cooking, and working immensely challenging. The strain on health and social care services is substantial, making the development of solutions that facilitate recovery outside of hospital settings, in the comfort of a patient’s own home, critically important.
How the Device Works
The Triceps trial centres on a small, non-invasive electrical device that is placed inside the ear. This device is designed to stimulate the vagus nerve, a major nerve that acts as a crucial communication pathway between the brain and the abdomen. Participants use this device concurrently with their prescribed rehabilitation exercises. The recovery process following a stroke is often protracted and demanding, requiring significant effort and countless hours of dedicated rehabilitation. "So we’re interested in ways we can boost the effects of rehab and create a brain environment that’s more responsive to it," Dr. Baig explains.
Unlike earlier iterations of vagus nerve stimulation, which necessitated surgical implantation of a device, this new treatment offers a non-invasive approach, enabling its use in the home environment. Dr. Baig assures that the electrical pulses delivered by the ear piece are calibrated to be comfortable and pain-free for the user.

‘A New Woman’
Amanda embraced the Triceps trial wholeheartedly, using the device for up to an hour each day as an integral part of her rehabilitation regimen over a 12-week period. This was meticulously combined with targeted exercises and the practice of everyday tasks. Initially, she harboured doubts: "At first, I didn’t think it was going to work at all." However, her persistence was rewarded. "But after a couple of weeks, I started to see changes in my hand."
One of the most significant milestones for Amanda was the ability to return to sewing, a beloved passion that had been rendered inaccessible in the wake of her stroke. The joy of this regained ability is palpable as she shares, "I feel like ‘a new woman’ now that I can cut material and use my sewing machine again." The impact extends far beyond her hobbies. "I can put my socks on, I can do my shoes, I can do my house," she exclaims, her voice filled with renewed independence. "I don’t have to wait for anyone else."
Early Improvements
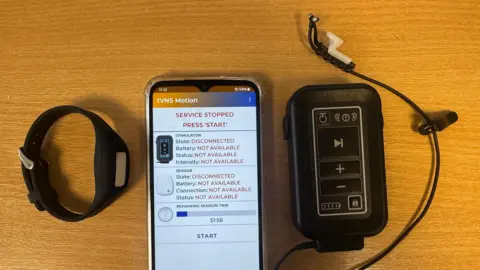
To date, the Triceps trial has seen over 200 individuals participate. While researchers are currently operating under a blind protocol, meaning they do not yet know which participants are receiving the active stimulation and which are receiving a placebo, the early indications are overwhelmingly positive. "We have seen some wonderful improvements in people’s arm function," Dr. Baig reports. He is careful to emphasize that this treatment is not a definitive cure but rather a tool that, if proven effective, could significantly enhance the quality of life for stroke survivors.
He illustrates the tangible benefits with compelling examples: "Somebody who was unable to carry a cup of tea with one arm can now walk from room to room holding it stably." Another participant achieved a personal best in a 5K run post-stroke, noting that their arm function had demonstrably improved during the activity.
The progress and findings of the Triceps trial are being closely observed by the Stroke Association, a key partner and part-funder of the research. Maeva May, the charity’s associate director of systems engagement, explains their keen interest: "The team are doing brain imaging and taking blood tests because we know that some stroke patients really respond well to this technology while some don’t." The ultimate goal is to unravel the underlying mechanisms: "We want to understand what patients respond best and how."
Dr. Baig remains optimistic about the potential scalability of this technology. He believes that if the trial yields positive results, the device could be "quite scalable" due to its affordability, user-friendliness, and its capacity for seamless integration into existing rehabilitation services.

For Amanda, the impact of this innovative technology is already profound and life-altering. "It’s about being independent again," she concludes, a sentiment that resonates deeply. "Those small things make a big difference." The hope is that this ‘at home’ approach to rehabilitation can empower countless other stroke survivors to reclaim their independence and significantly improve their daily lives.







