
The debate about whether the National Health Service (NHS) in the UK should embrace psychedelic-assisted therapy, specifically using psilocybin – the active compound in magic mushrooms – to treat debilitating mental health conditions like depression, is intensifying. This discussion sits at the intersection of promising scientific advancements, historical stigmas, and urgent public health needs. As conventional treatments often fall short for many, researchers and patient advocates are increasingly looking to a class of substances long relegated to the fringes of medicine.

Larissa Hope, a former actress who appeared in the TV drama Skins, stands as a testament to the potential transformative power of psilocybin. At 17, the sudden glare of fame unexpectedly unearthed buried trauma, plunging her into a severe mental health crisis. Traditional antidepressants offered little solace. It was a carefully administered dose of psilocybin, taken under strict clinical supervision, that marked a profound turning point. "When I experienced it, I burst out crying," she recalls. "It was the first time in my life I had ever felt a sense of belonging and safety in my body. I kept saying, ‘I’m home, I’m home’." Nearly two decades later, Larissa credits this single experience, alongside subsequent therapy, with helping her navigate and ultimately overcome suicidal ideation. Her story encapsulates the hope driving the current psychedelic renaissance.
However, not all experiences are positive, underscoring the complexities involved. Jules Evans, a university researcher, vividly remembers his first recreational LSD trip at 18, which spiralled into a terrifying "deluded" state. "I believed that everyone was talking about me, criticising me, judging me. I thought, I’ve permanently damaged myself; I’ve permanently lost my mind," he recounts. "It was the most terrifying experience of my life." Evans subsequently suffered from social anxiety, panic attacks, and was diagnosed with post-traumatic stress disorder (PTSD). Today, he directs the Challenging Psychedelic Experiences Project, dedicated to supporting individuals grappling with adverse effects from psychedelics. His work highlights the critical need for rigorous safety protocols, comprehensive screening, and robust therapeutic support to mitigate risks and manage potential harms.

These two starkly different narratives illustrate the core dilemma confronting doctors, regulators, and policymakers: how to harness the therapeutic potential of psychedelic drugs while safeguarding against their inherent risks. The burgeoning scientific evidence, particularly from the past decade, suggests that these substances could offer breakthrough treatments for conditions ranging from severe depression, obsessive-compulsive disorder (OCD), and PTSD, to trauma and various addictions including alcohol and gambling. The current legal framework in the UK, however, severely restricts their use to authorised research or clinical trials, categorising them as Schedule 1 drugs with "no medicinal value."
Since 2022, more than 20 clinical trials in the UK have been investigating different psychedelic medicines for these conditions. While many studies report promising results, showing significant reductions in symptoms and sustained improvements, others have yielded mixed or inconclusive findings, and a few have found no clear benefit on their primary outcome measures. A highly anticipated data release from one of the largest global clinical trials into psilocybin use, conducted by UK biotech firm Compass Pathways, is expected later this year. The UK’s medicines regulator, the Medicines and Healthcare products Regulatory Agency (MHRA), is keenly awaiting this data as it considers whether to ease existing restrictions and allow the medical use of psilocybin outside of research settings.

Prof Oliver Howes, chair of the Royal College of Psychiatrists’ Psychopharmacology Committee, expresses cautious optimism. He views psychedelics as a promising new frontier in psychiatric treatment, including for NHS patients. "One of the key messages is that this is something we desperately need – more treatments and better treatments for mental health disorders," he states. "These treatments are really interesting because they’ve shown promise in these small-scale studies… and have the potential to work quicker." However, he stresses the paramount importance of robust evidence, urging against "overhyping the potential benefits" before large-scale, definitive trial results are available. The Royal College of Psychiatrists itself issued a report in September 2025, cautioning about the potential dangers and stressing that recreational use is illegal and harmful.
The history of psychedelic use is as old as human civilization, with magic mushrooms, opium, and cannabis featuring in ancient rituals and recreational practices. In the mid-20th century, substances like LSD gained prominence, particularly within the 1960s counterculture movement, famously championed by Harvard psychologist Timothy Leary, who urged individuals to "turn on, tune in, drop out." However, this cultural association with social unrest and perceived moral decay led to a widespread ban by the late 1960s and early 1970s, effectively shutting down scientific research for decades.
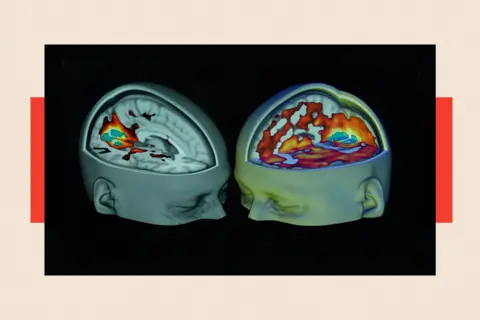
A turning point arrived in the 2010s with groundbreaking research by Prof David Nutt and his team at Imperial College London. Their work reignited scientific interest, demonstrating psilocybin’s potential to quickly "switch off" the part of the brain associated with depression, a stark contrast to the weeks required for conventional antidepressants to take effect, and often with fewer side effects. This faster onset of action is a significant advantage, potentially offering relief to patients who are unresponsive to current therapies. While Prof Nutt is a respected scientist, his outspoken views on drug policy, including controversial comparisons between the harm of ecstasy and horse-riding, led to his dismissal as chair of the government’s drugs advisory body in 2009. Nevertheless, his pioneering studies have inspired a global resurgence in psychedelic research.
At University College London, neuroscientist Dr Ravi Das is exploring how psychedelics might disrupt habitual behaviours linked to addiction. His study is recruiting heavy drinkers to investigate whether dimethyltryptamine (DMT), a short-acting psychedelic, can target the brain’s memory and learning systems, effectively "breaking down those associations" that reinforce addictive patterns. "Every time someone drinks, a bit like Pavlov’s dog, you’re learning to associate things in the environment with the rewarding effect of alcohol," he explains. If successful, this early-stage research could pave the way for NHS treatments, pending regulatory approval. Dr Das argues for equitable access: "If psychedelic therapies prove to be both safe and more effective than current treatments, I would hope to see them made accessible via the NHS – rather than to just the privileged few who can afford them privately."

Unlike psilocybin, LSD, DMT, and MDMA, which are Schedule 1 substances, ketamine occupies a different legal category and is already used in medical settings in the UK for certain conditions. Dr Das believes that mounting scientific evidence could compel the government to revise the scheduling of other psychedelics, easing the restrictive licensing requirements for research and clinical use. However, a November 2024 analysis in the British Medical Journal by PhD student Cédric Lemarchand and colleagues highlighted inherent challenges in evaluating psychedelic drugs. They noted the difficulty in disentangling the drug’s effects from the accompanying psychotherapy, complicating comprehensive evaluations and product labelling. The report also warned that short-term trials might not fully capture the "potential for harm and serious adverse events from long-term use of hallucinogens," and that "the potential for abuse or misuse must also be considered."
Despite the therapeutic promise, doctors like Prof Howes maintain that, outside of controlled research settings, psychedelic treatments should not become routine medical practice until larger, more rigorous trials provide definitive evidence of their safety and effectiveness. He warns against unsupervised use: "In a clinical trial setting, it’s very carefully evaluated. If people take these on their own or in a backstreet clinic, then there is no guarantee of that and the safety issues start becoming a major issue." Data from the Challenging Psychedelic Experiences Project corroborates these concerns, indicating that 52% of regular psychedelic users reported an intensely challenging trip, with 39% considering it among the five most difficult experiences of their lives. Furthermore, 6.7% considered self-harm or harming others, and 8.9% reported impairment lasting over a day. Some individuals required medical or psychiatric intervention, experiencing prolonged distress. Mr Evans insists that "ideally, I would love doctors and regulators to know more about these adverse effects, and how people can recover from them, before they say, any of these therapies are safe."

Yet, Prof Nutt, Prof Howes, and Dr Das universally agree that bureaucratic hurdles and the difficulty of obtaining research permissions are severely impeding progress. "There are so many people suffering unnecessarily," Prof Nutt lamented to BBC News, "And some of them are dying, because of the unreasonable barriers to research and treatment that we face in this country. It is, in my view, a moral failing." He argues that once proven safe and effective, these medicines must be available through the NHS, not solely confined to the private sector, drawing parallels with the limited access to medical cannabis. Prof Howes echoes this sentiment, urging the government to review regulations for research, acknowledging the "long delays" they cause in developing urgently needed treatments.
The analysis from Mr Lemarchand and his team calls for greater scrutiny of trials by medical journals, demanding critical appraisal, full disclosure of limitations, avoidance of "spin and unsubstantiated claims," and corrections where necessary. The Advisory Council on the Misuse of Drugs remains firm in its stance that Schedule 1 drugs, by definition, possess "no medicinal value" and thus warrant the tightest controls, a position supported by ministers who link the Home Office licensing regime directly to public protection.

While the government has indicated support for easing licensing requirements for some MHRA and Health Research Authority-approved clinical trials, with exemptions for certain universities and NHS sites in progress, the pace of change is slow. A cross-government working group is coordinating a cautious rollout, pending pilot project results. Prof Howes notes, "There’s still a lot of red tape holding things up."
Supporters of psychedelic medicines hold out hope that the results from Compass Pathways’ phase three trials will catalyze further regulatory relaxations, at least for research purposes. Larissa Hope, whose life was profoundly altered by psilocybin, firmly believes in the importance of these trials. Her experience provided crucial insight into her suicidal ideation and trauma, offering a path to healing where none seemed to exist. "I had a solid plan to end my life. Then suddenly, death wasn’t the only way," she concludes. "Under psilocybin, my nervous system began, for the first time, to recognise what peace felt like." The question for the NHS is not if, but when and how, it will grapple with this profound potential.






